12-Month Safety and Efficacy Evaluation of HORA-PDE6B, a Gene Therapy Targeting Patients with Retinitis Pigmentosa Due to Biallelic PDE6B Gene Mutation.
View Session View PresentationAdd to Schedule Print Abstract
Abstract Number: 2134
AuthorBlock: Jean-Baptiste Ducloyer1, Guylene Le Meur1, Pierre Lebranchu2, Fanny BILLAUD2, Lise Giraud2, Sandrine Meunier3, Nicole Brument3, Patricia Françon3, catherine MATHIS3, Serge Fitoussi3, Michel Weber1
1service d’ophtalmologie, TARGET, Université de Nantes, CHU Nantes, INSERM, NANTES, F-44000, France; 2Service d'Ophtalmologie, Université de Nantes, CHU Nantes, NANTES, F-44000, France; 3Clinical Development, Coave Therapeutics, PARIS, France;
DisclosureBlock: Jean-Baptiste Ducloyer, None; Guylene Le Meur, None; Pierre Lebranchu, None; Fanny BILLAUD, None; Lise Giraud, None; Sandrine Meunier, None; Nicole Brument, None; Patricia Françon, None; catherine MATHIS, None; Serge Fitoussi, None; Michel Weber, None;
Purpose
Retinitis Pigmentosa (RP) is a diverse hereditary disease leading to blindness after decades of irreversible progression. One form is associated with an autosomal recessive mutation in the Phosphodiesterase 6 Beta Gene (PDE6B). HORA-PDE6B is a clinical-stage gene therapy targeting RP patients with PDE6B mutations. It employs an Adeno-Associated Virus serotype 5 (AAV2/5) vector carrying the human PDE6B isoform 1 cDNA, controlled by the photoreceptor-specific human rhodopsin kinase promoter that targets Rods photoreceptors. A phase I/II study with HORA-PDE6B is currently underway, evaluating safety, efficacy, and immune response profiles. This work presents interim analysis results from the first 17 adult patients after 12 months of follow-up (FU).
Methods
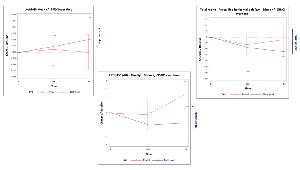
The treatment is given via subretinal injection in the worth seeing eye, with the fellow eye used as control. Patients are observed for five years, testing two dose levels: 3.4 x 1011 vg and 6.4 x 1011 vg per eye. Safety is assessed by Adverse Events (AEs)/ Serious Adverse Events (SAEs), while efficacy measures include Visual Field (VF), Visual Acuity (VA), Full-Field Stimulus Testing (FFST), Microperimetry, Mobility Test and Ellipsoid Zone (EZ) via Optical Coherence Tomography (OCT).
Results
Seven patients received the low dose, 10 got the high dose. Both doses were well tolerated, with 2 SAEs linked to HORA-PDE6 (one VA decrease and one chorioretinitis). The injection volume was reduced from 800 to 400 µl to mitigate the risk of retinal detachments reported in 2 out of 4 patients receiving 800 µl. Primary efficacy analysis was done in 5 patients at a less advanced disease stage receiving the higher dose. Treated eyes exhibited a favorable trend suggesting a potential therapeutic effect requiring confirmation through the coming 24-month data. For most parameters, the untreated eyes kept declining per disease's natural course, while stabilization occurred in most tests in the treated eyes (VA, VF, microperimetry, mobility test, and horizontal line of EZ). Blue light FFST improved, suggesting rods photoreceptors transduction cascade reactivation.
Conclusions
Over a short 12-month period, HORA-PDE6B has shown a favorable safety profile and potentially efficacy, encouraging the continued development of HORA-PDE6B.